Chasing Sunrises: New Hope for Autoimmune Arthritis Care
A 2026 roadmap exploring precision medicine, GZMK protein research, and the gut-joint axis in autoimmune arthritis.
The 2026 Roadmap: Precision Medicine and Cellular Breakthroughs
Autoimmune arthritis—once a condition of “managing the decline”—has entered the era of Immune Re-programming. For those of us navigating these chronic waters, the research coming out of early 2026 represents a fundamental shift in how we understand the “self-attack” of the immune system.
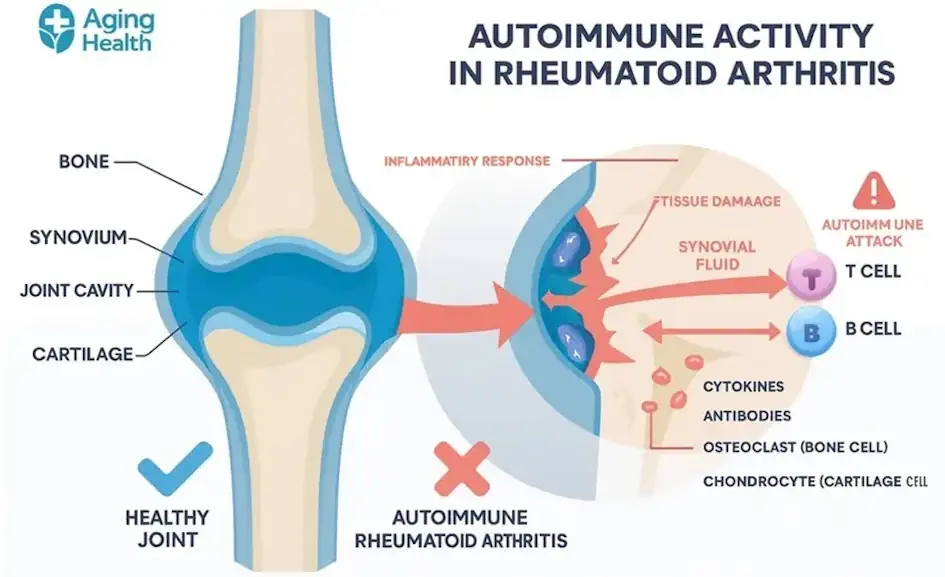 Figure 1: Infographic depicting the autoimmune process, emphasizing systemic inflammation and joint tissue damage.
Figure 1: Infographic depicting the autoimmune process, emphasizing systemic inflammation and joint tissue damage.
What is Autoimmune Arthritis in 2026?
It isn’t a single disease; it’s a systemic failure of Immune Tolerance. Unlike standard osteoarthritis (“wear and tear”), autoimmune varieties occur when the body’s defense force identifies synovial tissue as a foreign invader.
 Figure 2: Inflamed joint tissue in rheumatoid arthritis, with immune cells (in red and green) infiltrating the synovium, attacking the joint lining, and triggering swelling, pain, and cartilage damage. This close-up reveals the autoimmune assault underlying chronic joint destruction.
Figure 2: Inflamed joint tissue in rheumatoid arthritis, with immune cells (in red and green) infiltrating the synovium, attacking the joint lining, and triggering swelling, pain, and cartilage damage. This close-up reveals the autoimmune assault underlying chronic joint destruction.
The 2026 Research Frontier: Three Major Breakthroughs
- The GZMK Protein Discovery: Research from Mass General Brigham (Feb 2026) identified Granzyme K (GZMK) as the trigger that “tricks” the complement system into attacking healthy tissue. Blocking GZMK may allow us to stop joint damage without suppressing the entire immune system.
- RNA-Based Drug Synthesis: Scientists are now using RNA Therapy to “silence” specific genes. Instead of broad immunosuppressants, this therapy tells your cells to stop producing inflammatory proteins like TNF-alpha at the source.
- CAR-T Cellular Reprogramming: We are seeing the first clinical successes in “rebooting” the immune system by training T-cells to eliminate the B-cells that produce harmful autoantibodies.
Clinical Card: 2026 Biomarkers
| Biomarker | Definition | 2026 Clinical Relevance |
|---|---|---|
| GZMK Protein | The “Inflammation Trigger” | High levels indicate an active attack on joint tissue. |
| HLA-DRB1 | Genetic Shared Epitope | Accounts for roughly 60% of the genetic risk for RA. |
| ctDNA / RNA | Liquid Biopsy | Used to monitor drug efficacy at a genetic level. |
2026 Patient Advocacy Checklist
Hand this to your Rheumatologist at your next visit:
- GZMK/Complement: “Could GZMK be the driver of my persistent joint damage?”
- RNA Therapy: “Am I a candidate for targeted RNA-based gene silencing?”
- Biomarker Testing: “Can we use ctDNA testing to monitor my response to this biologic?”
- Microbiome: “How can we coordinate my treatment with gut-barrier integrity protocols?”
Precision Health FAQ: 2026 Update
1. Why is gut barrier integrity a priority for my arthritis? The Gut-Joint Axis is now a confirmed clinical pathway. A compromised gut lining allows microbial toxins (like Colibactin) into the bloodstream, which “primes” the immune system for a systemic attack on your joints.
2. What is the “10-Year Rule” for screening? For families with a history of early-onset conditions, screening should begin at age 40 or 10 years earlier than the age their first-degree relative was diagnosed—whichever comes first.
3. How does RNA-Silencing differ from Gene Editing? While CRISPR (Gene Editing) permanently changes DNA, RNA-Silencing simply “mutes” the instructions, “turning off” the disease proteins without altering your genetic code.
Related Patient Resources
- The Log: Track your metabolic metrics with the Daily Glucose Tracker.
- The Foundation: New to this? Read my Beginner’s Guide to Diabetes.
- Liver Health: How metabolic syndrome impacts liver cirrhosis.